
Patient-Centric Care: Continuity Beyond the Hospital Walls
January 25, 2026
Automated LabVIEW Testing for Accelerated CPAP/BiPAP Device Verification
March 4, 2026
Patient-Centric Care: Continuity Beyond the Hospital Walls
January 25, 2026
Automated LabVIEW Testing for Accelerated CPAP/BiPAP Device Verification
March 4, 2026
Implementing Test Automation Across the Medical Device Development Life Cycle for Medical Device Validation
Automated Testing Across the Medical Device Lifecycle
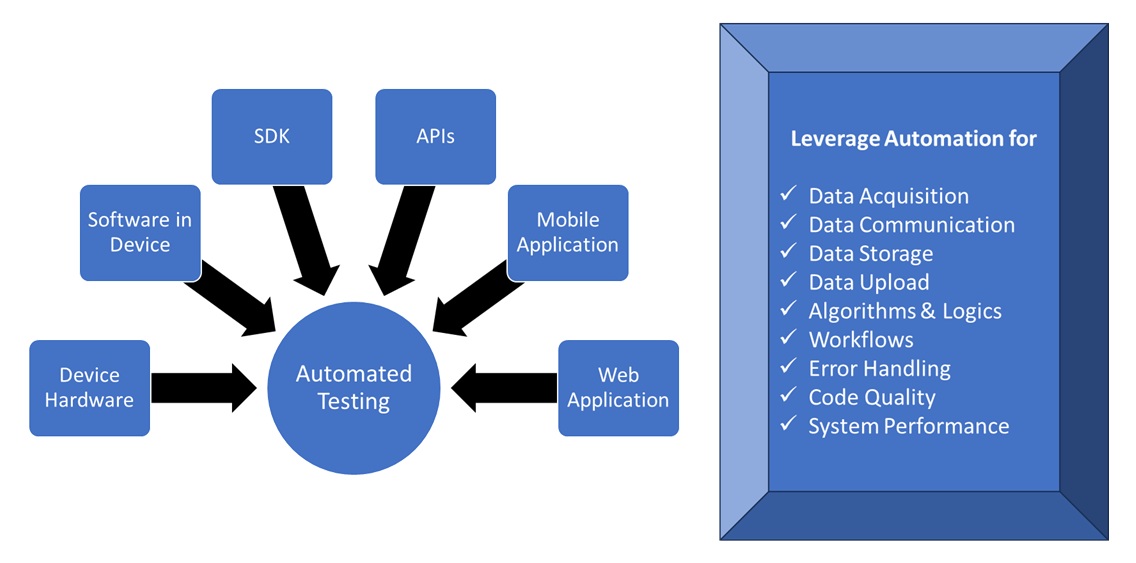
Patient safety, device reliability, and regulatory compliance necessitate comprehensive testing throughout the development, deployment, and post-market enhancement cycles of a medical device. Modern medical systems typically comprise device hardware and Software in a Medical Device (SiMD), ancillary software, Software as a Medical Device (SaMD), device interfacing applications, and cloud platforms, many of which integrate to deliver end-to-end medical device validation services. These activities collectively form the foundation of modern medical device software testing and structured healthcare application testing practices.
Verification and validation activities should therefore encompass technical, functional, nonfunctional, and integration testing across the device ecosystem, complemented by human-centred usability studies and clinical evaluations. To meet the demands of continuous improvement and regulatory oversight, testing must be rigorous, repeatable, and continuous. This level of assurance can be achieved through a comprehensive end to end test automation strategy, enabling faster cycles, traceability to requirements and early detection of issues, if any.
Signal Generators for Regression Testing
The use of physiological data simulators, such as iOrbit’s ECG Database Player, enables systematic verification and repeatable regression testing of algorithms and noise handling methods. The ECG player outputs multichannel analogue signals derived from diverse databases available on PhysioNet and other validated sources, allowing controlled and reproducible test conditions. This approach strengthens iOrbit’s medical device validation services by enabling repeatable algorithm verification across controlled physiological datasets. Also, it supports rigorous evaluation of algorithm stability, robustness against signal artifacts, and compliance with performance requirements across multiple datasets, thereby strengthening confidence in device reliability and regulatory readiness.
Device to SDK communication testing
Mock Bluetooth services are used to validate SDK APIs, ensuring that the Bluetooth stack and SDK functionality operate correctly even when physical hardware is unavailable. Automated testing covers connection handling, pairing, device status communication, and data transfer, enabling early verification prior to prototype readiness. At iOrbit, Bluetooth mocks combined with frameworks such as JUnit have been employed to simulate GATT services and characteristics, allowing thorough validation of SDK parsing, data integrity, propagation of processed data to the UI layer and error handling. This approach facilitates early defect identification and resolution at the SDK layer before integration with actual devices. Additionally, automated testing supports evaluation of multiple data configurations-such as varying sampling rates and signal resolutions-to confirm robust compatibility and performance across diverse device scenarios. Such automated verification represents an essential component of modern medical device software testing, particularly for connected healthcare ecosystems.
Automated testing through HL7/FHIR API data simulators
atient monitors and other similar devices expose FHIR / HL7 APIs to transmit patient data in a structured manner. In order to test the components integrated with such devices, automated data simulation scripts help in continuous streaming of patient data to interfacing systems to verify reliability and security of continuous data communication, performance and multi-device compatibility of interfacing components. Continuous data simulation supports large-scale healthcare application testing and validation of interoperable clinical systems.
Automated testing of iOrbit’s data collector app and gateway apps installed on the Hub device leverage the continuous data streaming through patient monitor simulator using HL7 structure. It helps in testing the integration of patient monitors with iTouch iOrbit’s cloud platform, performance testing of data collector app and sizing of the hub device to facilitate smooth communication.
Firmware automated testing using frameworks like Ceedling
Unit testing of device embedded software verifies the firmware logic in isolation, eliminating dependencies on real hardware by using mocks and stubs. Unit testing is conducted prior to deploying the code on real hardware. It helps in automation of peripheral driver logic such as GPIO, SPI, I2C, UART and ADC , middleware modules, application-level logic, state machines and utility functions like buffers, and error handling.
Different examples of iOrbit’s use of firmware automation test frameworks include testing of data processing and conversion logic (like sensor raw data processing, scaling, formatting), middleware logic used for filtering and safety checks, fault handling and recovery logic. It also includes testing for buffer management, CRC calculations, peripheral timeout scenarios, and state validation. Firmware-level automation plays a critical role in end-to-end medical device validation, ensuring embedded safety logic operates as intended.
Automated testing through patient simulators
Tools are leveraged to simulate patient’s physiological parameters and create a test structure leveraging these simulated datasets to verify the device response and accuracy. For example, patient monitors are tested by creating a automated test suite simulating ECG signal variations, arrhythmias, range of respiration rate, NIBP , and SpO2 values. The tests can be repeated over large duration of time in an automated manner to evaluate the monitor performance.
Similarly, testing of CPAP devices involves using pneumatic simulators to replicate patient’s breathing pattern, inhalation/exhalation cycles to validate device ability to maintain the airway pressure. Adding automated apnea simulation in breathing patterns helps in detecting CPAP device’s ability to apnea detection and alarm generation. These simulation-driven approaches also support Usability Risk Testing of Medical Devices, helping evaluate device responses under realistic patient interaction scenarios and strengthening overall safety validation.
Web API Test Automation
Integration of medical device software and ancillary applications with cloud servers typically relies on web APIs. Automated testing of these APIs, using tools such as Postman, enables early identification of issues related to functionality, security, and performance. For example, automated validation of patient admission and discharge APIs through positive, negative, and boundary test scenarios ensures proper authentication, secure communication, and correctness of workflow execution.
Incorporating API automation into the CI/CD pipeline provides continuous testing, enabling early defect detection and rapid feedback to developers. This approach enhances reliability, accelerates release cycles, and ensures traceability. API automation further strengthens healthcare app testing services, ensuring secure and reliable clinical data exchange across connected healthcare ecosystems.
Automated Static code analysis
iOrbit leverages tools like SonarCloud for continuous static code analysis to detect bugs, vulnerabilities, code smells, and maintainability issues, ensuring higher code quality and compliance with best coding practices. SonarCloud connects directly to GitHub repositories, with each build triggering static analysis and highlighting issues, if any. Continuous analysis contributes to scalable medical device validation services by ensuring maintainable, secure, and compliant software architecture aligned with regulatory expectations.
Software GUI-Based Automation
Automated testing of software workflows that integrate with medical devices enables faster, repeatable, and traceable verification of system stability and reliability. By leveraging multiple automation tools and frameworks such as Selenium, Appium, Maven, and TestNG, along with design approaches like Page Factory, data-driven testing, and integrated reporting utilities, iOrbit has established a robust testing methodology. This CI/CD integrated process ensures comprehensive coverage of user interface workflows, accelerates regression cycles, and provides traceability to requirements. Automated GUI workflows enhance healthcare application testing, validating usability, workflow integrity, and clinical user interaction. As a result, the testing process achieves greater efficiency, consistency, and confidence in the performance of medical device-integrated applications.
Automated Load and stress testing
The response behavior of web applications and server performance verification is conducted in iOrbit through performance testing tools like JMeter. It helps in simulating simultaneous requests from multiple users and API calls while measuring response times, throughput, and resource utilization under different load conditions. Performance testing forms an essential pillar of medical device software testing, ensuring scalability and system resilience under clinical workloads. It helps identify bottlenecks and scalability issues, enabling reliable deployment of connected healthcare systems.
Conclusion
A comprehensive test automation strategy enables organizations to deliver safe, compliant, and scalable healthcare solutions. By integrating medical device validation, healthcare application testing, and Usability Risk Testing of Medical Devices throughout the development lifecycle, manufacturers can accelerate innovation while maintaining regulatory confidence, system reliability, and patient safety.