
Bringing Real-Time Medical Device Data into Hospital Management System (HMS)
January 14, 2026
Transforming Care Delivery in Eldercare Homes with a Human-Centered Connected Care Solution
January 22, 2026
Bringing Real-Time Medical Device Data into Hospital Management System (HMS)
January 14, 2026
Transforming Care Delivery in Eldercare Homes with a Human-Centered Connected Care Solution
January 22, 2026From Concept to Care: End-to-End Product Engineering of a CPAP/BiPAP Respiratory Device

About Client
A major medical device manufacturer in Asia introducing indigenous sleep apnea respiratory devices with full design and manufacturing ownership, replacing an earlier OEM white-label CPAP/BiPAP device.
Client’s Challenge
To achieve complete ownership of CPAP/BiPAP product specifications and design post-transfer from iOrbit, enabling full-scale manufacturing and commercial release. The new custom CPAP/BiPAP respiratory device needed to deliver better performance than the existing OEM white-labelled product.
Key challenges included:
- Limited in-house resources bandwidth to design and develop a completely new cPAP/BiPAP machine
- Complex airflow dynamics and pressure control engineering
- Multi-mode operation (CPAP / APAP / BiPAP) within a single embedded system
- Enhanced usability engineering and human factors requirements
- Highly ergonomic and aesthetic industrial design (ID) for clinical and home use
- Complete Design History File (DHF) documentation ready for regulatory submissionsIEC/ISO tests to meet FDA consensus standards
iOrbit Approach / Solution
End-to-end CPAP/BiPAP medical device product development for a CE Class IIa respiratory device, covering design, validation, and regulatory readiness.
The solution included:
- Full electro-mechanical design with resolution of prior noise control issues observed in OEM devices
- Medical-grade companion app development for clinician-facing configuration and monitoring
- Compliant DHF documentation, cybersecurity risk management, safety risk management, and usability & human factors validation
- Alignment with global medical device certification standards
- Design implementation of the iBSM application
- Comprehensive risk management for product safety
- Extensive verification and validation (V&V) & generation of test reports. Design History File (DHF) documentation
- DHF, IFU labelling and Medical Device File readiness for submission leveraging the iOrbit 13485 processes
- Integrated system-level testing of the product.
- Design and Development of product workflows and protocols
- Completion of all statutory IEC/ISO tests to meet FDA consensus standards.
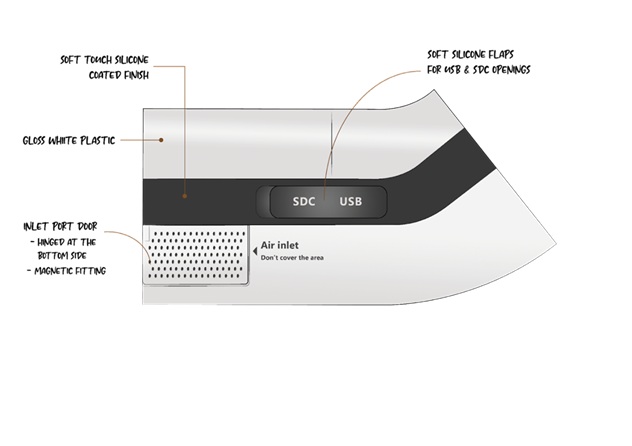

Key Outcomes
- Designed a CPAP/APAP/BiPAP respiratory product aligned with Product Requirement Specifications (PRS) and User Requirement Specifications (URS)
- Completed UX design and development of clinician-facing CPAP applications
- Achieved seamless integration of CPAP off-the-shelf (OTS) standard components
- Delivered mechanical and hardware design of an elegant and ergonomic device station
- Ensured product safety, risk management, and regulatory compliance
- Successfully completed verification & validation (V&V), usability, and human factors testing
- Delivered DHF, IFU, labeling, and regulatory documentation aligned with ISO 13485 process framework
Frequently Asked Questions
1What is involved in end-to-end CPAP/BiPAP medical device development?
End-to-end CPAP/BiPAP medical device development includes product definition, airflow and pressure control engineering, electro-mechanical design, embedded software, usability engineering, verification and validation, and regulatory-ready DHF documentation to enable safe commercialization.
2What regulatory standards apply to CPAP and BiPAP respiratory devices?
CPAP and BiPAP devices must comply with CE Class IIa classification, applicable IEC 60601 series standards, ISO 13485 quality management systems, usability standards, and FDA consensus standards for respiratory medical devices.
3What is a Design History File (DHF) for CPAP/BiPAP devices?
A Design History File (DHF) is a mandatory regulatory document that captures design inputs, outputs, risk management, verification, validation, usability studies, and change controls, ensuring compliance for CE marking and FDA submissions.
4How is airflow accuracy and noise control achieved in CPAP/BiPAP machines?
Airflow accuracy and noise control are achieved through advanced airflow dynamics modeling, precision pressure sensors, optimized blower selection, acoustic dampening, and closed-loop control algorithms validated during verification testing.
5Can OEM white-label CPAP/BiPAP devices be redesigned for full IP ownership?
Yes. OEM white-label CPAP/BiPAP devices can be re-engineered to achieve full IP ownership, improved performance, enhanced usability, and regulatory-ready design control, enabling independent manufacturing and market expansion.