
Automated LabVIEW Testing for Accelerated CPAP/BiPAP Device Verification
March 4, 2026
Automated LabVIEW Testing for Accelerated CPAP/BiPAP Device Verification
March 4, 2026Automated Medical Device Validation Using LabVIEW & NI DAQ for EMG-EIM Diagnostic Systems

Introduction to Medical Device Validation in Modern MedTech Systems
Modern diagnostic systems are becoming increasingly complex, requiring structured medical device validation, robust medical device software testing, and reliable healthcare application testing methodologies to ensure performance, safety, and regulatory compliance.
This article explores how LabVIEW and NI DAQ were implemented as an automated verification and validation framework for an EMG–EIM diagnostic platform, enabling repeatable testing, independent signal verification and regulatory-aligned validation workflows.
System Architecture for Medical Device Software Testing
Device Under Test (DUT)
The EMG–EIM system consists of:
- Analog Front End (AFE)
- FPGA processing modules
- Processing System (PS)
- PC-based validation environment
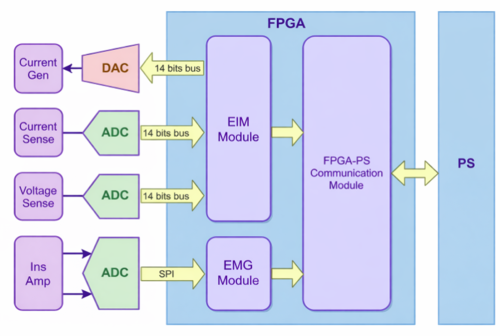
The validation setup interfaced with multiple signal nodes across the hardware and software stack, supporting comprehensive medical device software testing and system-level validation.
Using LabVIEW-controlled NI DAQ hardware, calibrated reference signals were injected into the AFE while internal processing outputs were captured for independent verification.
Automated validation frameworks should also align with regulatory expectations such as guidance from the FDA on the General Principles of Software Validation, ensuring medical device software is properly verified and validated before market release.
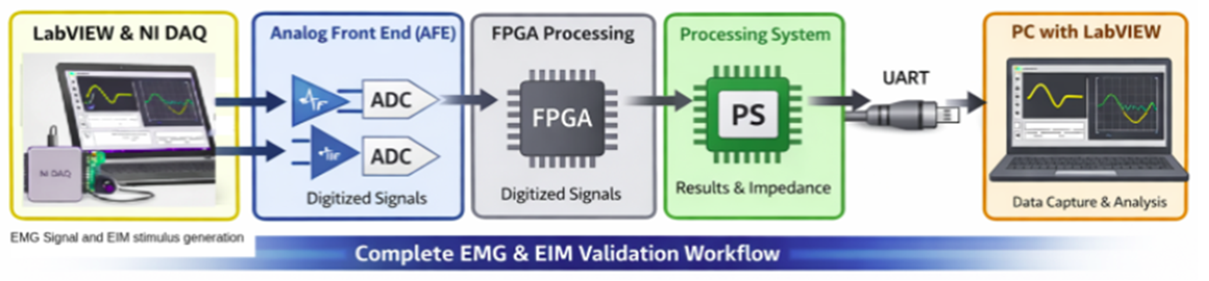
This architecture reflects modern medical device validation services practices used in regulated healthcare environments.
EMG Signal Verification & Validation - Healthcare Application Testing Approach
For EMG verification & validation:
- LabVIEW generated representative EMG waveforms.
- Signals were injected into the EMG instrumentation amplifier.
- The AFE digitized signals through ADC conversion.
- FPGA EMG algorithms processed incoming data.
- Processed data was transmitted via UART to the PC.
LabVIEW received data using VISA serial communication and performed independent analysis to validate:
- Signal integrity
- Gain accuracy
- Filtering performance
- Timing synchronization
- Noise characteristics
This workflow demonstrates practical healthcare application testing applied to physiological signal acquisition systems.
EIM Signal Validation - Automated Medical Device Validation Methodology
For Electrical Impedance Myography (EIM):
- NI DAQ generated controlled stimulus voltage and current signals.
- LabVIEW controlled amplitude, frequency and test sequencing.
- Validation included:
- Single-frequency Guide Mode
- Multi-frequency Analysis Mode
Captured signals were processed by FPGA algorithms and transferred to the Processing System for impedance computation.
LabVIEW independently validated:
- Impedance calculations
- Frequency response behaviour
- Signal stability
- Noise performance
This robust workflow demonstrates a practical and systematic approach to healthcare application testing, specifically applied to high-precision physiological signal acquisition and diagnostic MedTech systems.
UART Data Acquisition and Independent Software Verification
LabVIEW functioned as an external verification platform by:
- Receiving EMG and EIM data via UART using VISA communication.
- Decoding structured data packets.
- Reconstructing numeric datasets.
- Performing real-time visualization and analysis.
Separating the analysis environment from the device ensured unbiased verification - a key requirement in professional medical device software testing workflows.
Usability Risk Testing of Medical Devices Through Automated Validation
Beyond signal verification, automated testing supported Usability Risk Testing of Medical Devices by enabling:
- Repeatable clinical signal simulations
- Failure condition testing
- Performance boundary evaluation
- Algorithm robustness verification
Such validation reduces user-interaction risks and strengthens regulatory compliance readiness.
End-to-End Medical Device Validation Coverage
The LabVIEW + NI DAQ framework enabled complete verification coverage across:
- Analog acquisition chain
- FPGA signal processing
- Impedance algorithms
- Software data handling
- Communication interfaces
Automation improved traceability, repeatability and validation confidence - core deliverables expected from professional medical device validation services.
Scalable Healthcare Application Testing Strategy
Automated testing platforms can significantly accelerate medical device validation, improve medical device software testing efficiency, and enable scalable healthcare application testing for next-generation diagnostic systems.
A LabVIEW-based validation framework provides a reusable and structured testing environment that supports regulatory submissions, improves product reliability, and helps accelerate MedTech commercialization.
Conclusion
Automated validation frameworks using LabVIEW and NI DAQ provide a structured and scalable approach for verifying complex diagnostic systems such as EMG-EIM platforms.
By enabling automated signal generation, independent verification, and repeatable testing workflows, such frameworks significantly improve validation coverage across analog hardware, FPGA processing, and system-level software components.
This approach supports modern medical device validation practices, improves test traceability, and helps accelerate regulatory readiness for advanced MedTech products.
FAQ - Medical Device Validation Using LabVIEW & NI DAQ
1.What are the benefits of using LabVIEW for medical device validation?
Using LabVIEW for medical device validation enables automated test execution, real-time signal analysis, scalable test frameworks, and improved verification coverage across hardware and software components.
2. Why is LabVIEW commonly used in medical device testing?
LabVIEW provides a graphical programming environment that enables engineers to automate signal generation, data acquisition, and analysis, making it suitable for verification and validation of complex diagnostic devices.
3. How does NI DAQ support automated validation workflows?
NI DAQ hardware enables precise signal generation and high-accuracy data acquisition, allowing engineers to simulate physiological signals and validate system responses under controlled test conditions.