
Patient-Centric Care: Continuity Beyond the Hospital Walls
January 25, 2026
Implementing Test Automation Across the Medical Device Development Life Cycle for Medical Device Validation
March 3, 2026
Patient-Centric Care: Continuity Beyond the Hospital Walls
January 25, 2026
Implementing Test Automation Across the Medical Device Development Life Cycle for Medical Device Validation
March 3, 2026System Engineering and Firmware Development for a RF Ablation Unit

About Client
A leading medical technology company dedicated to improving women’s health aimed to develop an RF ablation system for controlled tissue ablation. The engagement focused on delivering medical device embedded software that could meet strict clinical and regulatory expectations.
Client’s Challenge
A new RF ablation hardware unit was developed with an Arduino-based prototype firmware. However, to progress toward commercialization, the device required medical device software development aligned with regulatory standards.
The client needed a complete firmware solution conforming to IEC 62304 medical device software requirements, meeting functional, safety, and fail-safe requirements, and fully aligned with the overall product architecture.
Project Objective
The primary goal was to create a robust, safe, and maintainable firmware architecture capable of real-time performance under stringent medical requirements. This included:
- Safe closed-loop ablation
- Deterministic response to sudden impedance changes
- Clear and intuitive user interface
- Robust safety shutdown mechanisms
- Easy field updates and long-term maintainability
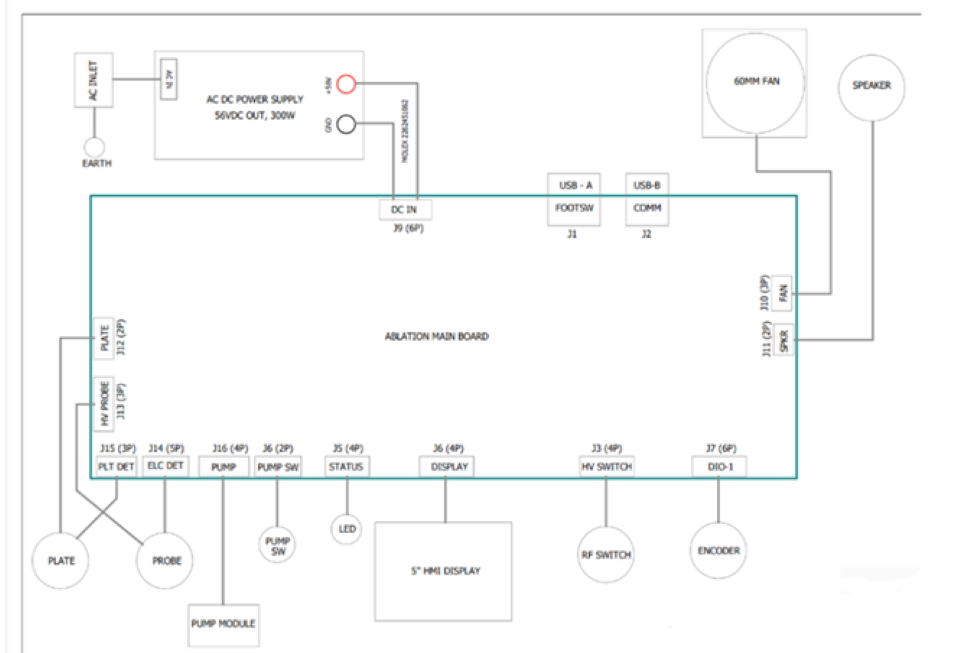
The device architecture includes a main ablation control board, pump module, RF switch, encoders, HMI display, audio alerts, fans, safety detection lines, and USB-based communication.
iOrbit Approach / Solution
We analyzed the complete hardware architecture to determine the optimal firmware development approach, tools, and design methodology. A scalable embedded firmware development strategy was selected to support real-time control, safety-critical operations, and future product extensions.
Key Highlights of RF Ablation unit Firmware:
The controller uses an ATmega2560 to manage:
- RF power control (via DAC → RF amplifier)
- Real-time impedance monitoring (HV probe + ADC)
- Pump & cooling control
- User interface (5" HMI, encoder, LEDs, footswitch)
- Safety monitoring, event logging, and USB communications
This architecture reflects best practices in medical device software development services for safety-critical systems.
Firmware Requirements
The firmware for the RF Ablation System provides reliable, real-time control of all device subsystems while ensuring patient safety, smooth user interaction, and deterministic response to sensor inputs. Built on the ATmega2560, the firmware must efficiently manage communication with peripheral components such as the pump module, RF switch, impedance sensing circuits, display, encoder, USB interface, and safety detection lines.
The system maintains stable operation during ablation, handles closed-loop RF power control, continuously monitors impedance and electrical conditions, and reacts instantly to abnormal events. It also supports a user-friendly interface, onboard data logging, built-in diagnostics, and a scalable architecture—key expectations in medical device embedded software projects.
Firmware Architecture Design
A multi-layered, modular architecture was developed to ensure clarity, testability, and compliance:
- Hardware Abstraction Layer (HAL)
- Device Drivers
- Application Layer
This structure supports long-term maintainability and simplifies verification validation and compliance activities.
Core Algorithms
- Impedance Measurement & real-time controlling
- Compute impedance using calibrated conversion.
- Maintain moving minimum/maximum values and short-term derivatives to detect sudden impedance spikes.
- Power Ramping & Closed-Loop Control (impedance driven)
- Ramp RF power in controlled cycles
- Ensure impedance stability during energy delivery
- Safety Engine
Faults continuously monitored include:
- Probe disconnect
- Overcurrent and overvoltage
- High impedance spikes
- Pump failure
- Thermal over-limit conditions
Safety logic was implemented in line with IEC 62304 medical device software risk-control requirements. - Probe disconnect
- State Machine
- The system transitions through INIT, SELTTEST, ABLATING, PAUSE, FAULT, and SHUTDOWN states to manage the complete workflow of RF ablation with safety and user control.
- Safety & Regulatory Considerations
- Deterministic response times for critical interrupts and fault lines
- Redundant safety checks: every action enabling high voltage requires both firmware validation and hardware interlock confirmation
- Design aligned with regulatory expectations for verification validation and compliance.
Key Outcome
- Scalable firmware architecture positioned for further development into a market-ready clinical product.
- Code reviews, unit testing, and system-level verification successfully completed
- Regulatory-aligned documentation supporting medical device software development services
Conclusion
The project concluded with the successful handover of fully evaluated firmware and system documentation. All functional, safety, and validation requirements were met, and the solution was delivered ready for further clinical and regulatory progression.