
Implementing Test Automation Across the Medical Device Development Life Cycle for Medical Device Validation
March 3, 2026
Automated Medical Device Validation Using LabVIEW & NI DAQ for EMG-EIM Diagnostic Systems
March 12, 2026
Implementing Test Automation Across the Medical Device Development Life Cycle for Medical Device Validation
March 3, 2026
Automated Medical Device Validation Using LabVIEW & NI DAQ for EMG-EIM Diagnostic Systems
March 12, 2026Automated LabVIEW Testing for Accelerated CPAP/BiPAP Device Verification

About Client
A major medical device manufacturer in Asia introducing indigenous sleep apnea respiratory devices with full design and manufacturing ownership, replacing an earlier OEM white-label CPAP/BiPAP device.
Client Challenge
Developing and validating a CPAP/BiPAP respiratory device requires rigorous verification of safety-critical components such as pressure sensing, blower motor control, and alarm mechanisms. The client needed to ensure accurate pressure delivery, reliable motor performance, and proper system response to fault conditions to meet strict regulatory and patient safety requirements.
However, their existing validation approach relied largely on manual and semi-manual testing, making it difficult to achieve comprehensive test coverage, repeatability, and objective pass/fail evaluation. Testing complex scenarios—such as pressure accuracy across operating ranges, motor response under varying conditions, and fault-handling behavior—was time-consuming and challenging to document in a traceable manner for regulatory audits.
The client required a structured automated testing framework that could systematically validate device performance, simulate real-world and fault conditions, and generate reliable, traceable validation data aligned with medical device regulatory expectations.
iORBIT Solution Overview
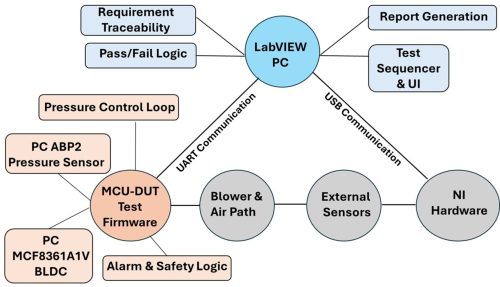
iORBIT adopted a practical approach to medical device validation and software testing through a structured Verification & Validation (V&V) and automated testing architecture for a CPAP/BiPAP device, using LabVIEW as the test executive
The architecture demonstrates how structured healthcare application testing and automated verification frameworks can improve compliance, safety, and performance validation in regulated MedTech environments.
System Setup
Device Under Test (DUT)
The CPAP/BiPAP device under test consists of a blower driven by a BLDC motor, a closed-loop pressure control system, and comprehensive safety alarms with fault-handling mechanisms. Internally, the system uses I²C communication exclusively for device-level interfaces, where the pressure sensor and the BLDC motor controller communicate with the MCU.
An external UART interface is provided between the MCU and the LabVIEW test PC for test control, telemetry, diagnostics, and medical device software testing activities.
The MCU operates as the sole I²C master, ensuring deterministic communication, controlled fault injection, and an auditable system architecture suitable for medical device validation services, regulatory documentation, and structured healthcare application testing services.
LabVIEW-Based Automated Test Architecture
The LabVIEW-based automated test architecture enables scalable medical device validation, traceable test execution, and regulatory-aligned documentation for CPAP/BiPAP systems.
This structured automation approach supports both functional testing and usability risk testing of medical devices, ensuring that system responses, alarms, and safety mechanisms behave as intended under real-world and fault conditions.
Automated Test Coverage and Verification Method for Mission-Critical Component – Pressure Sensor
The automated verification of the pressure sensor covers accuracy and linearity across the operating pressure range, noise and resolution under steady-state conditions, drift behaviour over extended operation, and response to over-range and saturation scenarios. In addition, fault detection and corresponding alarm responses are verified.
For verification, LabVIEW compares pressure values acquired from an external reference pressure sensor through the NI DAQ against the internal pressure values reported by the MCU over UART. Based on predefined acceptance criteria, LabVIEW automatically evaluates the results and generates objective pass/fail decisions, with all raw data and outcomes logged for traceability.
This structured automation strengthens medical device validation services by ensuring objective data comparison, measurable acceptance criteria, and audit-ready reporting-all essential for compliant healthcare application testing services.
Automated Test Coverage and Verification Method for Mission-Critical Component - Blower Motor
The automated verification of the BLDC motor controller focuses on evaluating blower and motor performance under closed-loop pressure control. This includes verification of motor speed behaviour as a function of commanded pressure setpoints, and assessment of pressure ramp response and settling characteristics.
In addition, fault handling and recovery behaviour are verified by inducing representative motor and load fault conditions through test firmware and observing system response.
LabVIEW acquires motor RPM and fault status via the MCU over UART, correlates these parameters with externally measured pressure and flow where applicable, and automatically evaluates the results against predefined acceptance criteria to generate objective pass/fail outcomes with full data traceability.
Such automation enhances medical device software testing, strengthens medical device validation, and contributes to comprehensive usability risk testing of medical devices, particularly for life-support respiratory systems.
Summary
Using a LabVIEW-based automated testing framework with a UART test interface and internal I²C communication, iOrbit established a robust and scalable architecture for validating CPAP/BiPAP respiratory devices.
This approach enabled precise and repeatable verification of critical subsystems such as pressure sensing, blower motor control, alarm functions, and safety mechanisms while maintaining clear system boundaries and traceable test outcomes.
The automated framework strengthened verification and validation (V&V) by generating objective pass/fail results, improving test coverage, and producing audit-ready documentation. As a result, the client benefited from improved regulatory readiness, higher product reliability, and greater confidence in device performance and patient safety for modern respiratory care systems.